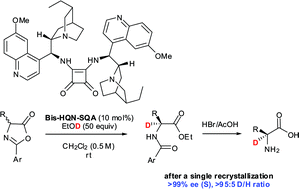
Cobalt‐Catalyzed Asymmetric Deuteration of α‐Amidoacrylates for Stereoselective Synthesis of α,β‐Dideuterated α‐Amino Acids - Li - 2023 - Angewandte Chemie International Edition - Wiley Online Library

Synthesis of Enantioenriched α-Deuterated α-Amino Acids Enabled by an Organophotocatalytic Radical Approach | Organic Letters
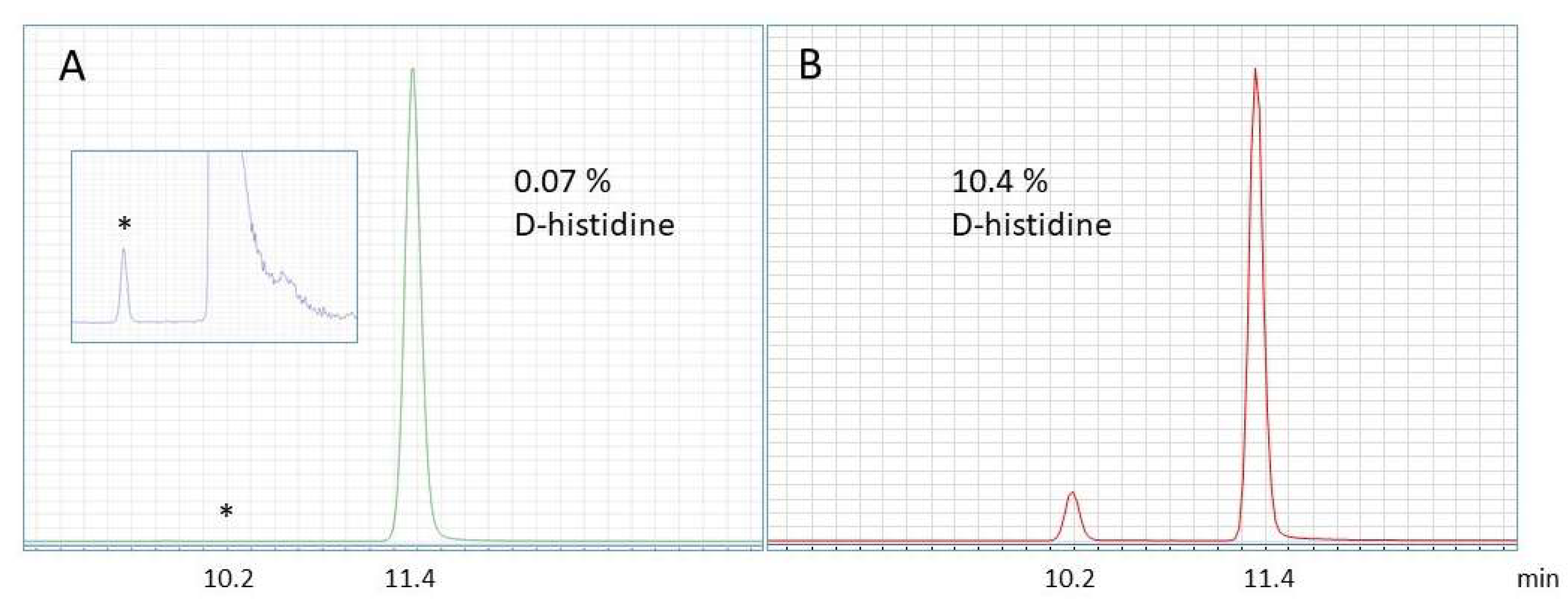
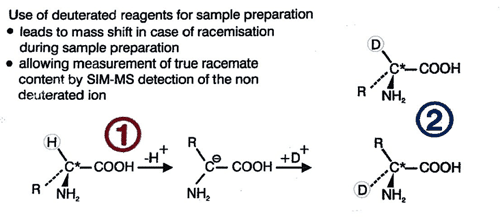
Foods | Free Full-Text | Simultaneous Determination of L- and D-Amino Acids in Proteins: A Sensitive Method Using Hydrolysis in Deuterated Acid and Liquid Chromatography–Tandem Mass Spectrometry Analysis

Synthesis of Enantioenriched α-Deuterated α-Amino Acids Enabled by an Organophotocatalytic Radical Approach | Organic Letters

Synthesis of Enantioenriched α-Deuterated α-Amino Acids Enabled by an Organophotocatalytic Radical Approach. - Abstract - Europe PMC
Schematic illustration of changes in the zero-point vibrational energy... | Download Scientific Diagram

Synthesis of Enantioenriched α-Deuterated α-Amino Acids Enabled by an Organophotocatalytic Radical Approach. - Abstract - Europe PMC

Identification of endogenous acyl amino acids based on a targeted lipidomics approach - ScienceDirect

Synthesis of β-Deuterated Amino Acids via Palladium-Catalyzed H/D Exchange | The Journal of Organic Chemistry

Synthesis of Enantioenriched α-Deuterated α-Amino Acids Enabled by an Organophotocatalytic Radical Approach | Organic Letters

H–D exchange in deuterated trifluoroacetic acid via ligand-directed NHC–palladium catalysis: a powerful method for deuteration of aromatic ketones, amides, and amino acids - ScienceDirect
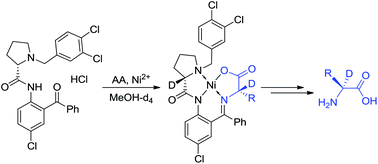
Asymmetric synthesis of α-deuterated α-amino acids - Organic & Biomolecular Chemistry (RSC Publishing)

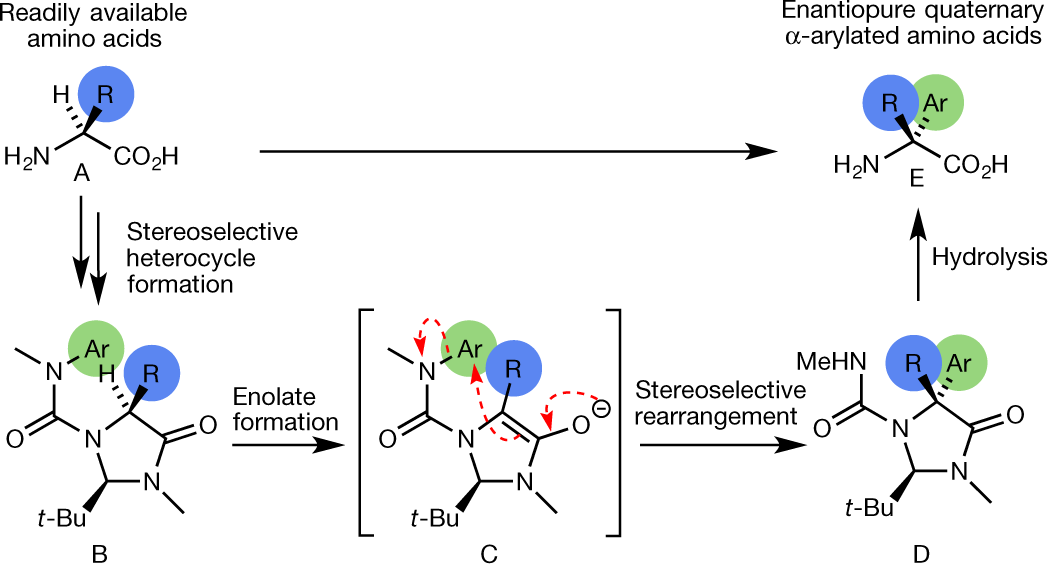
Expedited synthesis of α-amino acids by single-step enantioselective α-amination of carboxylic acids | Nature Synthesis
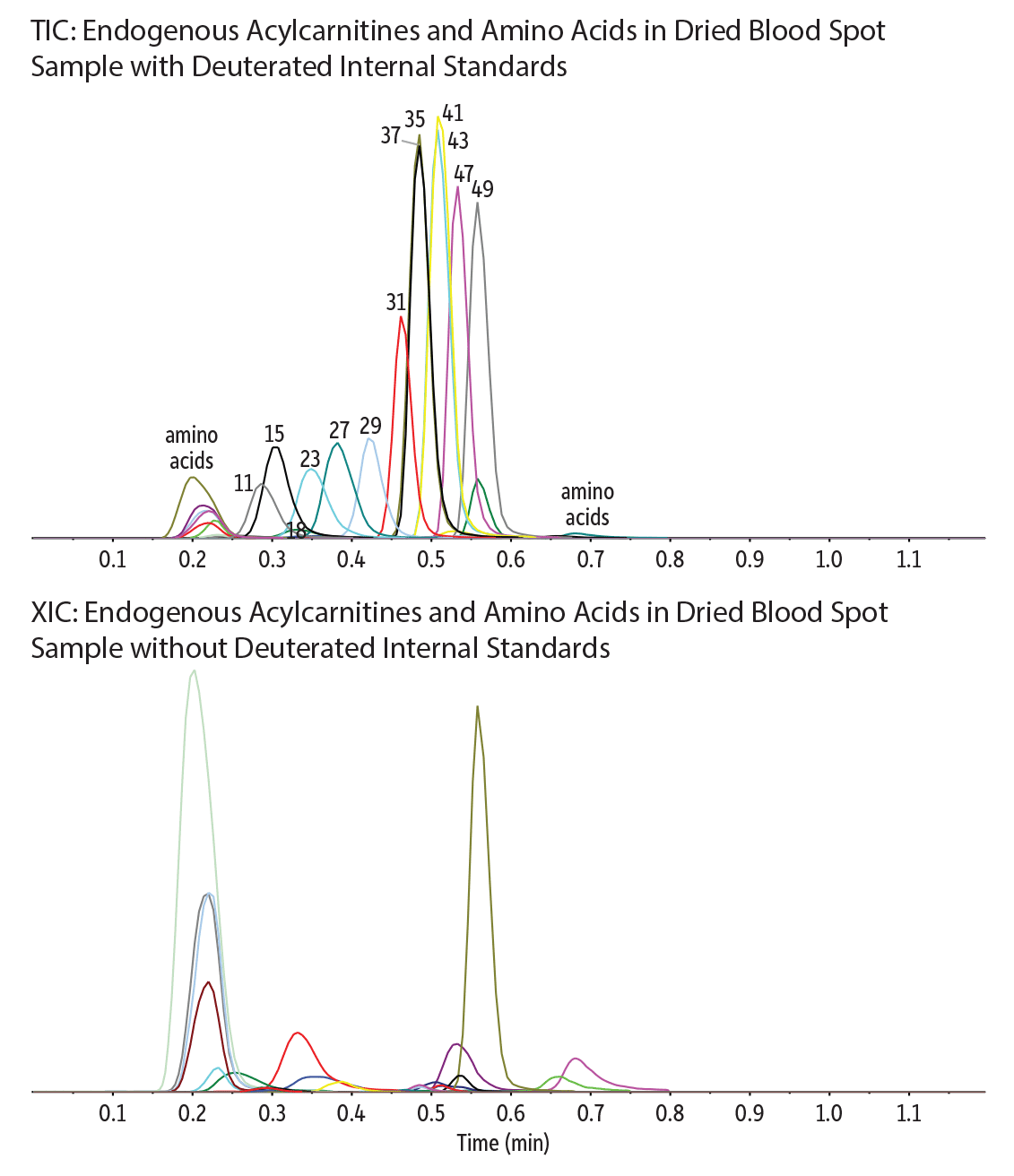
Restek - 22 Acylcarnitines and 13 Amino acids (Endogenous) in Dried Blood Spots with 16 Deuterated Internal Standards on Raptor HILIC-Si EXP Guard Cartridge Column by LC-MS/MS

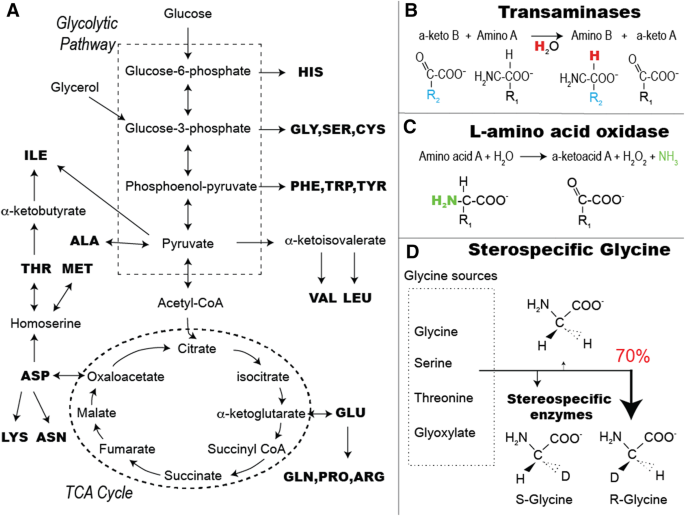
Site-Selective Deuteration of Amino Acids through Dual-Protein Catalysis | Journal of the American Chemical Society