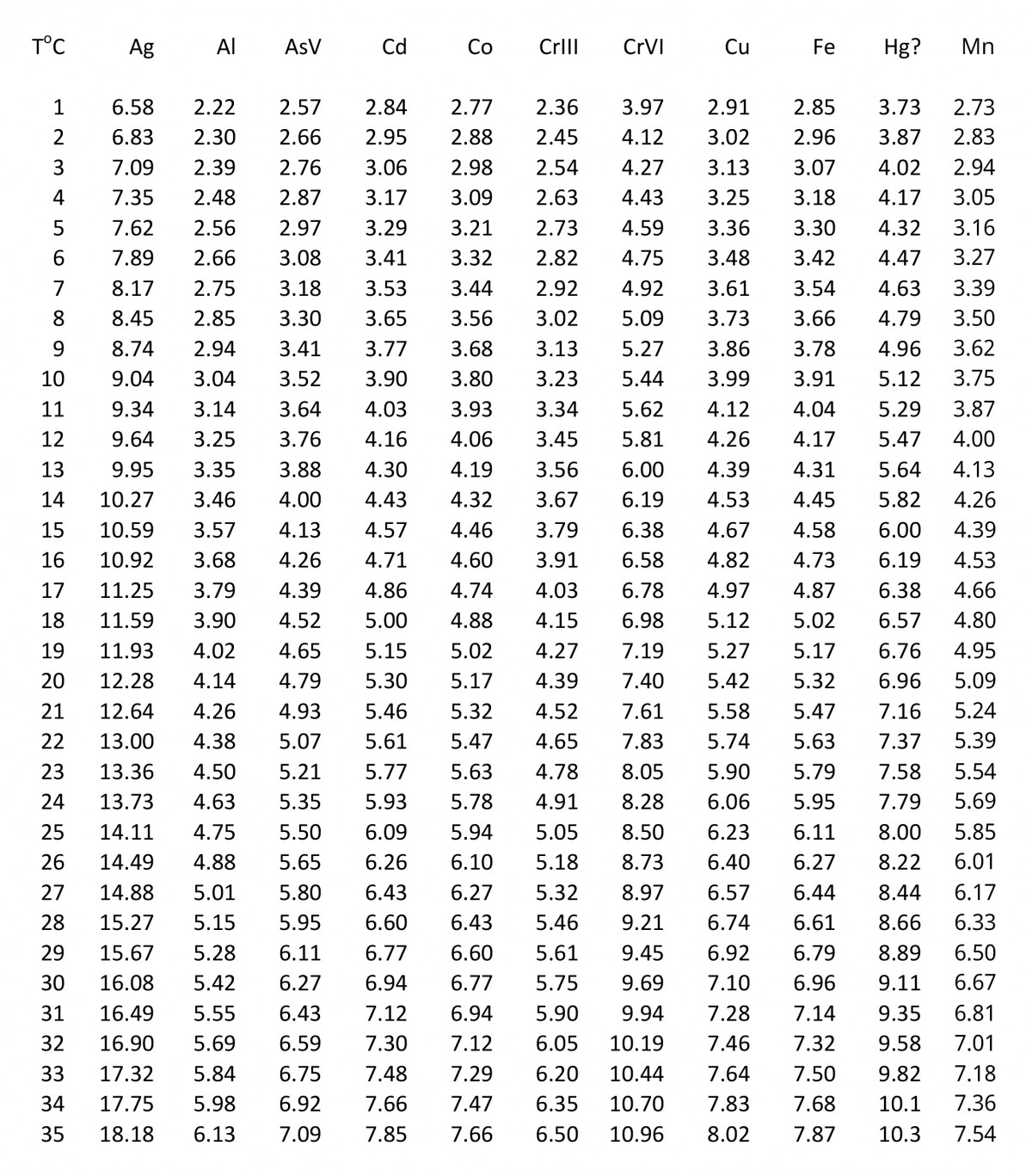
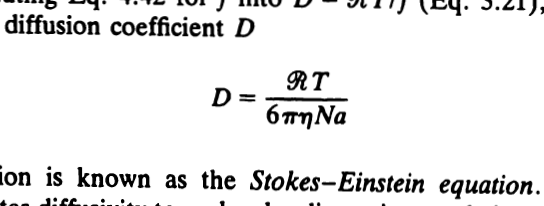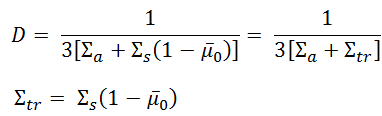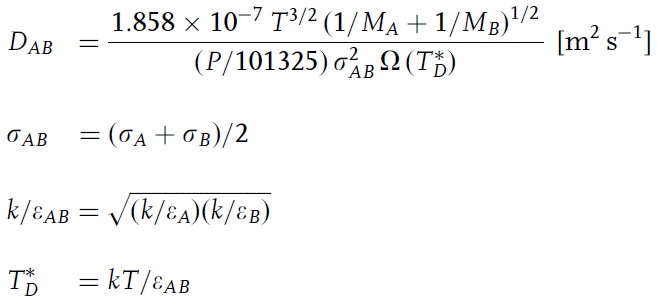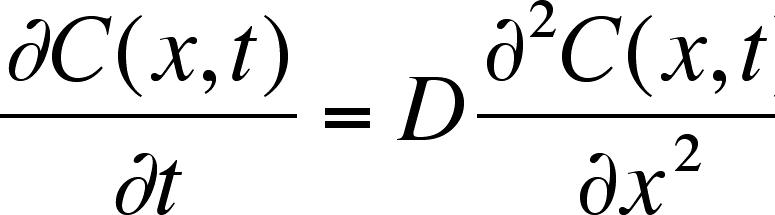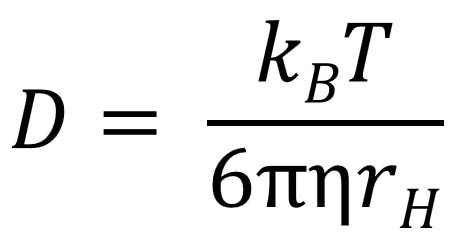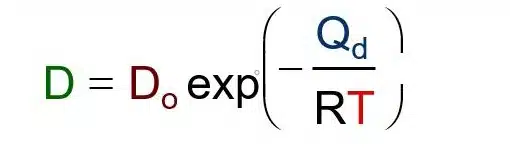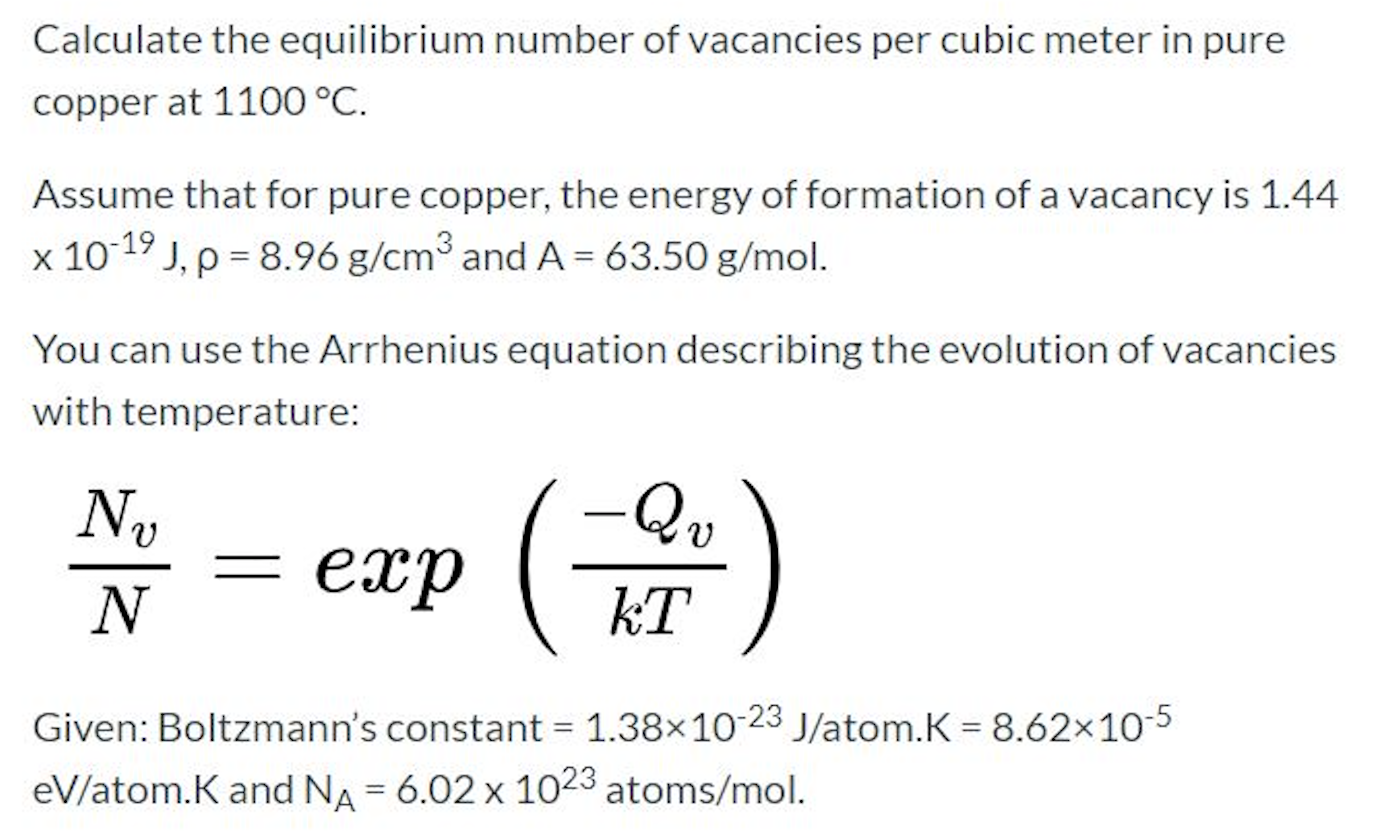Simple and accurate correlations for diffusion coefficients of solutes in liquids and supercritical fluids over wide ranges of temperature and density - ScienceDirect
ACP - Viscosities, diffusion coefficients, and mixing times of intrinsic fluorescent organic molecules in brown limonene secondary organic aerosol and tests of the Stokes–Einstein equation

SOLVED: According to the Kinetic Theory of gases, the diffusion coefficient; D, of a gas can be described using the following equation: 1 D = v^ 3 Using this ratio calculates the

The diffusion coefficient of an ideal gas is proportional to its mean free path and mean speed. The absolute temperature of an ideal gas is increased 4 times and its pressure is